Sour Skittles are addictive.
In an attempt to enjoy all of the tongue-tingling flavor I could, I once held each individual Skittle on my tongue until all of its sugary sour coating had dissolved. This was a great idea at first. After a while though, my tongue started to burn. I powered through, intent on maximizing my Sour Skittles experience. But by the time I finished the pack, my tongue was completely raw. Did I taste the rainbow? No. Actually, I couldn’t taste anything for the next two days.
Luckily for me, the cells on your tongue regrow quickly so no harm, no foul. But what happened? Citric acid—the same stuff that gives lemons their mouth-puckering sour flavor—makes up that delicious coating on the outside of Sour Skittles. And that acid began to eat away at my tongue. While I was able to handle a little bit of the citric acid, I overexposed myself. Unfortunately, my Sour Skittles experience has parallels with an overlooked aspect of climate change—ocean acidification.

Ocean acidification has been referred to as climate change’s “equally evil twin” but gains far fewer headlines than climate change itself. Despite its lack of notoriety, it could have far-reaching consequences—ones far worse than not being able to taste things for a couple of days.
According to a 2013 study, our world’s oceans absorbed an average of 2.5 petagrams (that’s the same as aquadrillion grams) of carbon—about half the weight of Lake Michigan—each year between 2002 and 2011. And this only accounted for about a quarter of all carbon dioxide (CO2) emitted over the same time period. For comparison, 2.5 petagrams of carbon dioxide is about the same amount that is absorbed by land-based plants. It’s also equal to the weight of 41 trillion bags of Sour Skittles.
Without the ocean, we would already be seeing much higher concentrations of CO2 in our atmosphere, as well as the accompanying stronger impacts of climate change.
“The ocean is just amazing in terms of dampening the effects of what we would feel with climate change. So, because of its absorption of carbon and heat from the atmosphere, we’re feeling the effects of climate change less than we would without its role,” says Natalie Freeman, a PhD candidate in the Atmospheric and Oceanic Sciences department who studies ocean acidification.
The ability of a liquid to absorb a gas like CO2 depends on the gas’ concentration. More gas in the air means more gas dissolving into the ocean. Therefore, as the level of CO2 in our atmosphere continues to rise through fossil fuel burning, the amount taken in by our oceans rises hand-in-hand.
The ocean absorbing CO2 from the air seems like a good thing.
However, this buffer is not without its own adverse consequences. “When CO2 gets taken up by the ocean, it goes through all these chemical reactions, leading to more hydrogen ions,” Freeman says. More specifically, the dissolved CO2 reacts with water to produce bicarbonate. These bicarbonate molecules react further to produce hydrogen ions and subsequently raise the acidity of the ocean.
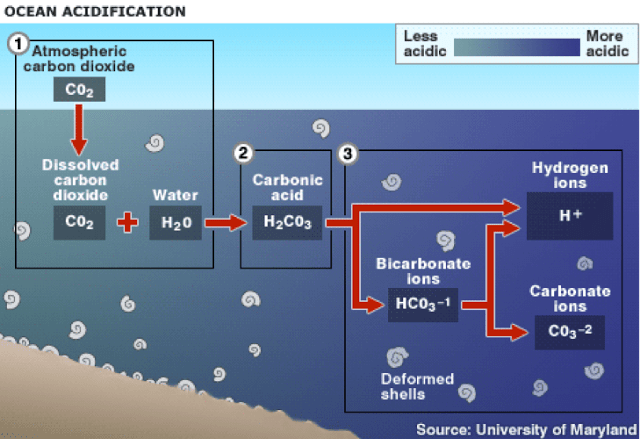
Since the industrial revolution and the subsequent proliferation of fossil fuel burning, the pH of the ocean has fallen from 8.2 to 8.1 (Pacific Marine Environmental Laboratory—PMEL). Because pH works on a log scale, this difference represents about a 30% increase in acidity.
This dramatic change in acidity results in creatures like corals and phytoplankton (tiny photosynthetic organisms) struggling to develop their skeletons and shells. This may not seem important to us but animals like these are the base of the marine food chain. Changes to phytoplankton may affect the cod and tuna we eat and the fishing communities which rely on marine life.
How else will ocean acidification affect us land-dwelling humans? More on ocean acidification and Skittles next Wednesday.
By Ryan Harp
