Flu outbreaks have already spread in many US states, predominated by a strain that tends to produce more severe symptoms. The flu vaccine has also been less effective this year due to a mutation that occurred during vaccine production. All this is causing experts to worry that this flu season might be worse than usual. As we brace ourselves for this winter’s flu, researchers at CU Boulder have discovered a new way that our cells fight off flu infection.
The flu is caused by an influenza virus that invades the nose, throat, or lungs of its human host. Once inside the cells of its host, the job of the virus is two-fold: evade the body’s defense system, and make copies of itself that can then infect neighboring cells, and eventually neighboring humans. One of the main gate-keepers that these viruses must thwart to achieve both goals, according to a new paper in Cell Host and Microbe, is a protein called TRIM25.

Influenza virus, the causative agent of flu
In the past decade, scientists have identified TRIM25 as a key activator of the immune response. TRIM25 helps trigger immune molecules called interferons to mount an attack on invading pathogens. Researchers at CU have now shown that TRIM25 can also prevent influenza viruses from making copies of their genome. They show that TRIM25 acts as a molecular clamp to hold viral proteins and RNA in place, making it more difficult for the virus to make copies of its genome.
Dr. Nicholas Meyerson, a post-doc in Dr. Sara Sawyer’s lab at CU Boulder, was the primary author on this study. Meyerson is an evolutionary virologist, which means that his research focuses on host-virus interactions and how these interactions change over evolutionary time.
“We work under the theme of a host-virus arms race in the lab, where we like to think of interactions between a host and the virus, and the pressure that that interaction puts on humans,” said Meyerson. He highlighted that the conflicting goals of virus spread and host health drive many of these evolutionary changes.
In the Sawyer lab, Meyerson uses genetic data from primates to find areas of the genome that are variable between species with common ancestors. These rapidly evolving areas often code for proteins that interact with pathogens. He then uses virology and biochemistry lab techniques to manipulate these proteins in cells to understand how the observed genetic differences affect how the protein interacts with viruses.
In fact, the novel antiviral activity of TRIM25 was discovered in just this way: an evolutionary screen that Dr. Sara Sawyer did as a postdoc detected many rapidly-evolving genes. When Meyerson started working in her lab, he says, “We had no idea what TRIM25 did. We were simply interested in it because of an evolutionary signal.” A year after starting this research into TRIM25, a paper was published in Cell Host and Microbe demonstrating that the flu virus binds to TRIM25 and prevents it from activating the host immune response.
While this paper validated some of Meyerson’s suspicions about TRIM25, it left many more questions open. The Cell Host and Microbe paper showed that the power of TRIM25 to activate the immune response was responsible for its ability to crush infection. However, Meyerson found that it could also stop viral infection without activating the immune response. This was when he started thinking that TRIM25 may play more than one role during influenza infection.
In this study Meyerson and his colleagues use primate versions of TRIM25 to understand how this protein is able to activate the immune response and also prevent viral replication.
“When you’re looking for novel antiviral proteins, the human version of the protein isn’t the best one to study, because the virus knows how to get around [human proteins] and they tend to be totally ineffective,” said Meyerson. “Viruses that infect humans aren’t adapted to the immunity proteins in other species.”
“The other convenient thing is that [primate] versions of these proteins are … almost identical to the human versions,” Meyerson continued. “So when you find differences in activity [they] tend to map to a small number of amino acid differences.”
With this “monkey business”, Meyerson and his colleagues showed that TRIM25 blocks the flu virus before the immune response would have time to kick in. They were also able to show that TRIM25 binds particles called viral ribonucleoproteins (vRNPs). These complex particles are made up of a segment of the viral genome wrapped around a protein core and bound by the polymerase, the same polymerase that makes copies of the virus’s genome. They were even able to demonstrate that this binding prevents the genome from being copied, thus halting the flu life cycle.

TRIM25 was already known to be a key activator of the immune response by generating Ubiquitin chains. Meyerson discovered a second anti-viral action.
The final piece of data that really convinced Meyerson and Sawyer that they had found a true separate function for TRIM25 were the electron microscopy images that showed purified viral ribonucleoproteins bound to TRIM25. According to Meyerson, the picture was worth a million words. “To actually see TRIM25 binding these elongated vRNPs … it took for me to finally see that, I think, to become a believer in my own theories.”
I have to agree with Meyerson on this one. The microscopy is a nice visual touch to the story he had been uncovering all along. Thanks to a dedicated team of researchers, we now know how special TRIM25 is, both in activating our immune response and in suppressing the ability of viruses to make copies of themselves. Meyerson and his colleagues have certainly shed light on a very interesting part of our body’s battle with influenza that could provide leads for new antiviral development.
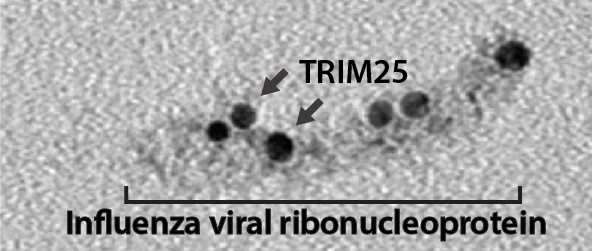
Electron microscopy showing TRIM25 binding to the influenza virus ribonucleoprotein
For now, Meyerson is looking more broadly at the evolution of this protein and trying to further characterize how it interacts molecularly with the flu viral ribonucleoproteins. His work serves as a reminder that there is still much to be learned about how our bodies interact with the pathogens around us.
As this flu season kicks into full gear, I also want to remind you of the great research that has already been done in the field of influenza, particularly in vaccine development. Vaccines help our bodies produce antibodies for surface proteins of the predicted seasonal flu strains. Remember that even though the flu vaccine is based on predicted flu outbreak data and is on average only 30-60% effective, your vaccination is part of a larger movement to greatly reduce the flu risk for children and the elderly who are most at risk of death from the flu.
By Kelsie Anson
Sources:
https://www.cdc.gov/mmwr/volumes/66/wr/mm6648a2.htm?s_cid=mm6648a2_w
http://www.cell.com/cell-host-microbe/pdfExtended/S1931-3128(17)30438-9
http://www.cell.com/cell-host-microbe/abstract/S1931-3128(09)00107-3
