A human cell’s set of defenses against infection can be seen as a type of custom security system. On a scale ranging from the loose deadbolt on your apartment door to the fingerprint scanners, motion detectors, and laser fields that I am led to believe are used in all casinos, bank vaults, and fancy museums, the human cell ranks right near the top.
Unfortunately, though, bacteria have developed clever tricks for getting around the cell’s security system and causing infections. In order to fight these infections, researchers at the University of Colorado have developed a new technique to find out how bacteria are picking our security system locks.
When the bacteria Listeria monocytogenes infect a human cell, they overcome cell defenses using a set of virulence factors. Virulence factors are small molecules and proteins which perform a variety of functions during infection. The phase of the infection, from invasion to replication to escape, determines when the bacteria secretes a particular virulence factor and where they go in the host.
The functions and carefully coordinated roles of these virulence factors, within the grand scheme of the infection, can be understood as a set of characters in a summer blockbuster spy/heist/action movie. Is this the best way to understand them? Probably not. But this is a thought experiment that I believe must be conducted, for the sake of scientific rigor.

How are listeria like our favorite action movie stars?
The first tools used by Listeria monocytogenes in this movie are the adhesion factors. These virulence factors are Nicholas Cage in National Treasure with fake fingerprints, or Tom Cruise in Minority Report with a fake eyeball. They engage with the outer surface of a mammalian host cell, fooling the cell into letting the bacterium inside.
Next are the virulence factors responsible for cell invasion. These factors together are the cunning mastermind whose ingenious plan hinges on getting captured.* They get the host cell to rearrange its own structural components (its cytoskeleton) and bring the bacterium inside. The cell will try to keep the invader contained in an acidic bubble, hoping to eventually destroy it. However, before that can happen more virulence factors stage a daring breakout by rupturing the compartment’s membrane, allowing the bacterium to break free from its intracellular prison bubble.
Throughout the infection, more secreted virulence factors let the bacteria hijack and overcome different defense mechanisms in the host, evading the security systems and drastically weakening the cell from the inside. This allows the rag-tag team of misfit Listeria bacteria to pull off a heroic escape and ride triumphant into the sunset/neighboring cell. Roll credits.

Triumphant listeria at the end of their heist movie? (Nope – image credit to Ocean’s 11)
Researchers all over the world are trying to make sense of the chaos caused by listeria infection and determine what each of these individual virulence factors is doing. Some of the biggest hints they get come from looking at where the proteins go inside in the host cell. For example, we know that the virulence protein called LLO deactivates host “alarm bell” genes. The team that discovered this function was given a major clue when they discovered that LLO localizes to the host cell nucleus, where those genes can be turned on or off.
A new technique out of Professor Amy Palmer’s lab in the Biochemistry department at the University of Colorado, Boulder, seeks to take this protein surveillance to a new level. They are able to look not only at where bacterial protein goes within a host cell, but when it goes there.
Visualizing protein location in a cell is not particularly new concept, as microscopes have been able to detect this for years. However, for gram-positive bacteria (a broad classification of bacteria) like Listeria monocytogenes, determining where a certain bacterial protein goes during different points of infection has been limited to particular pre-determined time points, rather than a complete movie. This is because the cells have to be “fixed”, or frozen in time in order for the protein-staining dyes—dyes that allow microscopes to see the proteins in the first place—to be added.
“Depending on what time point you’re looking at, you see the localization of the protein but you really don’t get the dynamic aspect,” said Dilara Batan. Batan, a graduate student at CU, took the lead on developing this technique as an undergrad in the Palmer lab.
Now, in any good heist movie, one of the key elements to infiltrating the high security vault is cutting the surveillance camera footage, disabling all efforts to monitor the place. Prior to this new technique, researchers were not exactly working with totally cut-off surveillance footage, since these fluorescence-based methods were available. However, rather than getting a full video of the bacteria heist, one would have to pick a point in time—say 12 hours into the heist**—to take a picture of the vault and see what information they could get from that.
Batan spent much of her undergrad research career putting together this method that gets those disabled surveillance cameras back online. Her work surveys dynamic protein behavior by building on the split green fluorescent protein (GFP) system. This system allows the bright green protein to be broken into fragments that give off green light only when joined together again.
In Batan’s research, one small GFP fragment is attached to the end of a bacterial virulence protein, while the rest of the protein is added to the host cell cytoplasm. She hypothesized that when that protein enters the cytoplasm to do its job in the heist/spy mission/evil plan, the GFP fragment tail would find the complementary GFP fragments in the host cell and start to fluoresce, letting her observe the localization dynamics throughout the whole infection, no fixing necessary.

Left: the blue virulence factor has a piece of GFP attached to it. Only when both pieces are combined (right) will the GFP glow green. Via Batan et al.
In previous research, the Palmer lab used a similar technique to monitor infections by gram-negative bacteria, but until now it remained unclear whether this method would translate to a gram-positive system.
“The gram-negative secretion systems are more well-understood than the gram-positive [secretion systems],” said Batan. “So we had no idea if it was going to work at all.”
Luckily, Batan’s hypothesis was correct. She tagged a bacterial protein—charmingly called InlC—and was able to watch it accumulate in a single cell over an 18-hour experiment.
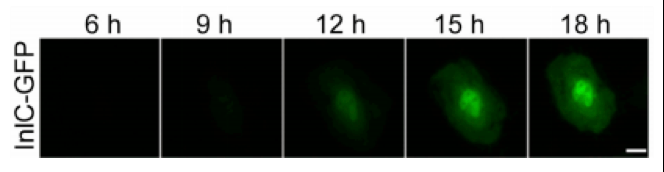
lnlC tagged with GFP accumulates in a single human cell over the course of 18 hours. Via Batan et al.
Now Batan (and others) can test whether individual cells in a population of host cells all acted the same during infection, and whether the bacteria behaved differently in different host cells. Previously, because you had to fix an infected cell to study it, there was no way to know what how a cell would change throughout infection.
Though she no longer works in the Palmer lab, Batan believes they will continue to explore technologies that improve our understanding of the ways bacteria are able to circumvent our state-of-the-art security system (which the salesman assured us was unbreachable, though he did seem a little shady in hindsight). These technologies help us figure out ways to exploit the weaknesses in bacteria’s masterplans and fill in the holes of our immune response that pathogens use to their advantage. For once, we want this heist movie to end without the ragtag squad of misfits escaping, because we’re the ones being cheated!
By Tom Aunins
*Examples: George Clooney in Ocean’s Eleven, Heath Ledger’s Joker in The Dark Knight, Javier Bardem’s Raoul Silva in Skyfall, Loki in the Avengers, and (I guess) Benedict Cumberbatch as Khan in Star Trek: Into Darkness, even though by 2013 most people (read: me) were pretty over this twist.
**For the sake of argument, let’s assume this heist is going in super slo-mo
Sources
https://www.tandfonline.com/doi/pdf/10.4161/viru.2.5.17703?needAccess=true
https://pubs.acs.org/doi/abs/10.1021/ja994421w?journalCode=jacsat

[…] listeria bacteria was published in Biophysical Journal. Also be sure to check out Tom Aunin’s article for Science Buffs about Dilara’s paper and also how listeria recreates your favorite heist […]
LikeLike